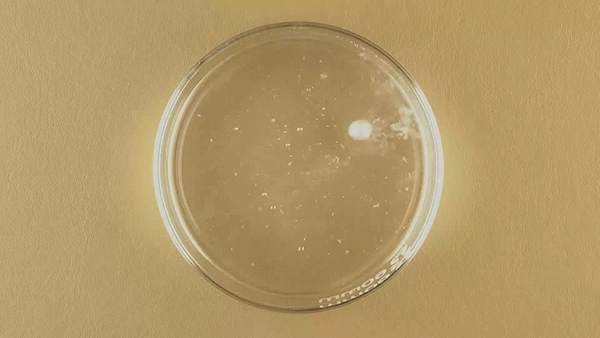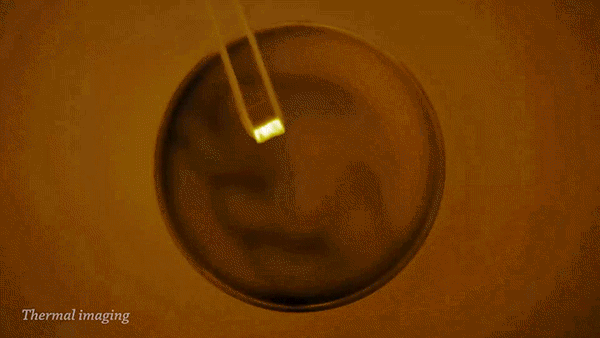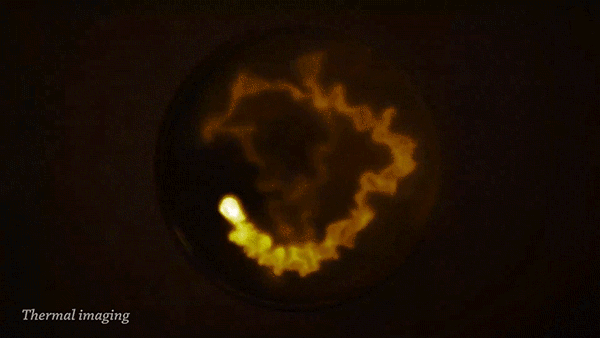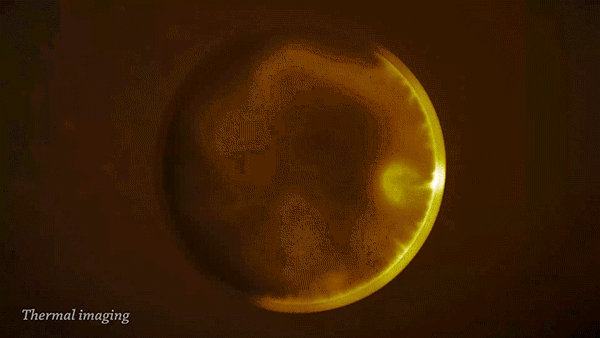
The reaction of sodium (Na) with water (H2O) is strongly exothermic and forms a colorless solution of sodium hydroxide (NaOH) and hydrogen gas (H2).
2Na + 2H2O -> 2NaOH + H2
During the reaction, the sodium can become so hot it will catch on fire and ignite the flammable hydrogen gas. This can be clearly seen when imaged with a thermal camera.
Source: https://redd.it/86cplg (u/mtimetraveller)
#ScienceGIF #Science #GIF #Sodium #Water #Chemical #Reaction #Thermal #Imaging #Exothermic #Chemistry #Melt #Heat #Hydrogen #Gas View Original Post on Google+